Our Pipeline

Through our proprietary Spyglass platform and focus on ecDNA biology, we have identified three distinct vulnerabilities of oncogene amplified cancers: (1) DNA Segregation (2) Replication Stress, and (3) DNA Assembly and Repair.
DNA Segregation: BBI-940
ecDNA lack centromeres, the structural component of chromosomes required for proper segregation of DNA during cell division (mitosis). Thus, segregation and subsequent inheritance of acentric chromosomal DNA, including ecDNA, into dividing cancer cells is dependent on alternative mitotic mechanisms. We have identified a novel kinesin target (“Kinesin”) essential to ecDNA segregation and inheritance in cancer cells, but non-essential in healthy cells. Inhibition or degradation of Kinesin is synthetic lethal to certain chromosomally unstable (CIN) and ecDNA-enabled cancer cells.
BBI-940 is a novel, oral, potent, selective degrader of Kinesin that has demonstrated robust anti-tumor activity across a range of cancer cell lines and mouse xenograft models, including single agent tumor regressions. In February 2026, we initiated our phase 1, first-in-human clinical trial of BBI-940 in patients with estrogen receptor positive and human epidermal growth factor receptor 2 negative, or ER+/HER2-, breast cancer as well as patients with triple-negative breast cancer luminal androgen receptor subtype, or TNBC-LAR. We refer to this trial as KOMODO-1 (Kinesin Oral Molecular Degrader for Oncology-1).
For patients and caregivers: to learn more about the KOMODO-1 study, please visit ClinicalTrials.gov Study Identifier NCT05827614.
Replication Stress: CHK1
Cancer cells with high copy number gene amplifications, including on ecDNA, exhibit elevated levels of replication stress (RS), a cellular condition where the DNA replication process is dysregulated and leads to potentially toxic DNA damage to cells. As a consequence, tumor cells with high copy number gene amplifications undergoing elevated RS invoke certain cellular RS response factors, exposing a synthetic lethal vulnerability, which renders them hypersensitive to inhibition of such factors.
Checkpoint kinase 1 (CHK1) is one of cancer cells’ master regulators of the DNA Damage Response to RS. We developed both preclinical and clinical CHK1 inhibitors with exceptional potency and selectivity and demonstrated single agent and combination potential against oncogene amplified cancers.
DNA Assembly and Repair: RNR
In order to maintain and perpetuate gene amplifications, including those on ecDNA, gene amplified cancer cells have increased demand for the raw materials used for DNA synthesis and repair. As a consequence, disruption of the supply of raw materials necessary for DNA assembly and repair can sensitize gene amplified cancer cells, including those that rely on ecDNA.
Ribonucleotide reductase (RNR) is the rate-limiting enzyme essential for de novo synthesis of deoxyribonucleotide triphosphates (dNTPs), the building blocks of DNA required for its assembly and repair. Inhibition of RNR increases RS and is synthetic lethal to cancer cells that rely on gene amplification for survival, such as those leveraging ecDNA amplification as a primary driver or as a resistance mechanism to overcome treatment. We discovered selective RNR inhibitors that effectively prevent and treat ecDNA-mediated acquired resistance to KRAS inhibitors. Amplifications are recognized as a major mechanism of acquired resistance to both selective and pan-KRAS inhibitors. Combinations with selective non-myelosuppressive RNRi constitute a therapeutic opportunity in cancers with resistance amplifications.
New ecDNA Targets and Vulnerabilities
We utilize Spyglass to identify and interrogate drug targets that exploit cellular vulnerabilities of oncogene amplified cancers. Our target identification efforts have revealed multiple distinct nodes of vulnerability within the lifecycle of ecDNA. We continuously incorporate new models, tools, and technologies into our Spyglass platform to interrogate CIN and ecDNA biology. In addition to our programs described above, we have preclinically validated multiple additional targets and have historically initiated ecDTx drug discovery efforts to identify potential candidates against such targets. We continue to deploy Spyglass to inform the development of BBI-940 and potential complementary targets or assets that we may acquire or internally develop in the future.
Spyglass: the only platform built to identify vulnerabilities of ecDNA in cancer
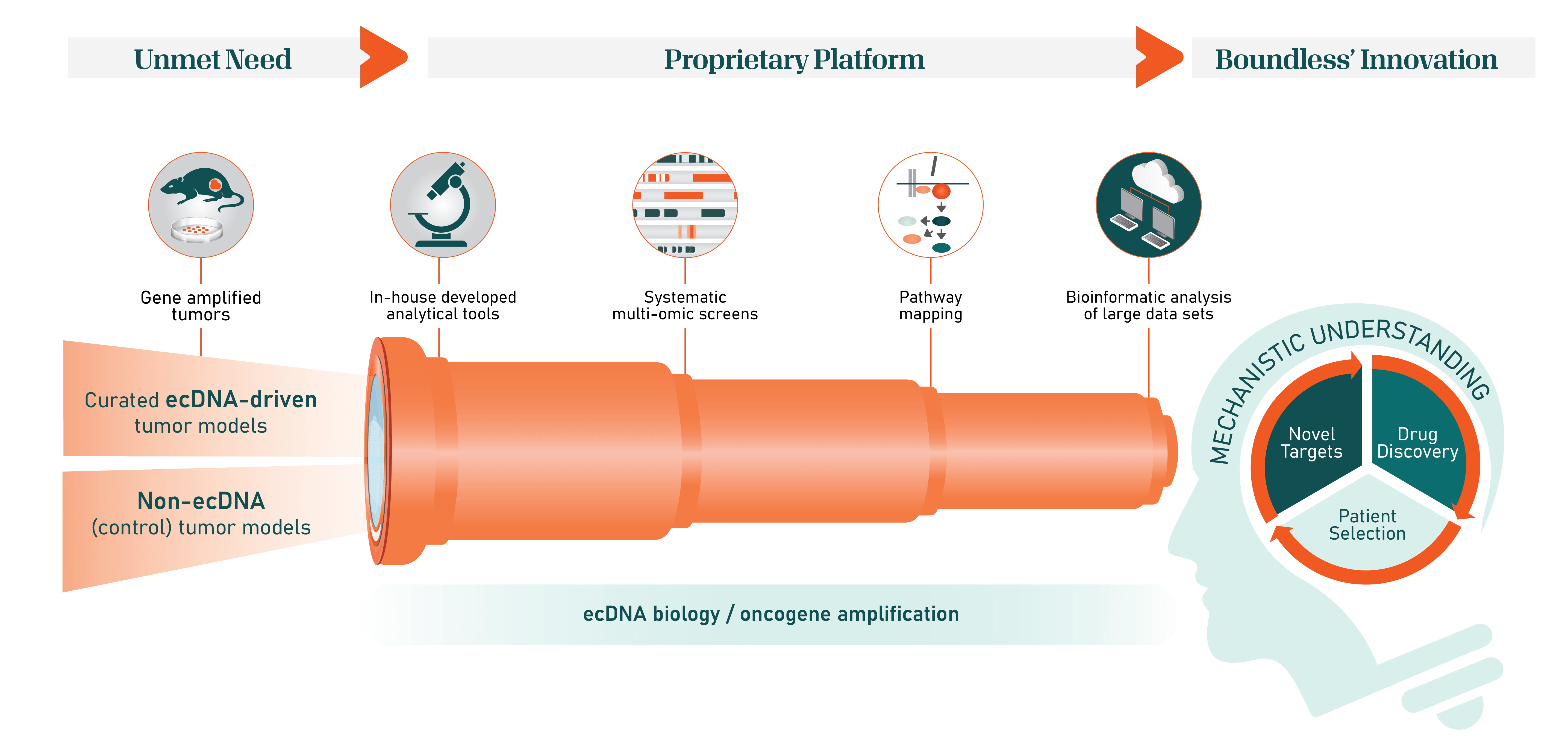
The Spyglass platform consists of a comprehensive suite of proprietary ecDNA+/- models that span many tumor types and amplified oncogenes. Leveraging innovative molecular analytical technologies and imaging techniques, Spyglass enables characterization of ecDNA in cancer cells and reveals a synthetic lethality-based approach to targeting ecDNA-enabled cancers. Through Spyglass, Boundless Bio scientists have thus far identified and validated several distinct and druggable cellular targets that are essential to the function of ecDNA in cancer cells.
ecDNA are the next frontier of precision medicine

ECHO (ecDNA Harboring Oncogenes) is a proprietary software algorithm for detecting the presence of ecDNA using routine clinical next-generation sequencing (NGS) data. ECHO is the first ecDNA diagnostic used in clinical trials.
In our mission to deliver transformative precision therapies to patients with oncogene amplified cancers, ECHO is a vital tool to select appropriate patients for treatment with our ground-breaking ecDTx candidates.
Foundational
Publications
Reviews
Examples of unmet need in
oncogene amplified cancer
Biomarkers predict enhanced clinical outcomes with afatinib versus methotrexate in patients with second-line recurrent and/or metastatic head and neck cancer
COHEN 2017 ANNALS OF ONCOLOGY
ReadFutibatinib, an Irreversible FGFR1-4 Inhibitor, in Patients with Advanced Solid Tumors Harboring FGF/ FGFR Aberrations: A Phase I Dose-Expansion Study
MERIC-BERNSTAM 2022 CANCER DISCOVERY
ReadProgression-Free Survival Among Patients With Well-Differentiated or Dedifferentiated Liposarcoma Treated With CDK4 Inhibitor Palbociclib: A Phase 2 Clinical Trial
DICKSON 2016 JAMA ONCOLOGY
ReadThe rapidly growing field of ecDNA cancer biology helps explain why some oncogene amplified cancers are so aggressive and why traditional approaches to treatment are not working for many patients. This new understanding of ecDNA shines light onto innovative and differentiated therapeutic approaches to help those affected by the most aggressive tumors. I am thrilled that Boundless Bio has assembled a world class team that has been able to leverage ecDNA biology to develop a host of innovative drug candidates that have the chance to improve the lives of patients with oncogene amplified cancer.”
Paul Mischel, M.D.
Scientific Co-Founder and Chairman of the Scientific Advisory Board

